利用者:加藤勝憲/骨形成タンパク質 4
An Error has occurred retrieving Wikidata item for infoboxAn Error has occurred retrieving Wikidata item for infobox骨形成タンパク質4は、ヒトではBMP4遺伝子によってコードされているタンパク質である[1][2]。BMP4は染色体14q22-q23に存在する。
Bone morphogenetic protein 4 is a protein that in humans is encoded by BMP4 gene.[1][2] BMP4 is found on chromosome 14q22-q23.
BMP4は、トランスフォーミング成長因子βスーパーファミリーに属する骨形成タンパク質ファミリーのメンバーである。スーパーファミリーには、成長因子や分化因子の大きなファミリーが含まれる。BMP4は進化的に高度に保存されている。BMP4は初期胚発生において、腹側辺縁帯、眼球、心臓の血液、耳小胞に存在する[3]。
BMP4 is a member of the bone morphogenetic protein family which is part of the transforming growth factor-beta superfamily. The superfamily includes large families of growth and differentiation factors. BMP4 is highly conserved evolutionarily. BMP4 is found in early embryonic development in the ventral marginal zone and in the eye, heart blood and otic vesicle.[3]
Discovery
[編集]骨形態形成タンパク質はもともと、脱灰骨抽出物がin vivoで骨格外の部位に軟骨内骨形成を誘導する能力によって同定された。
Bone morphogenetic proteins were originally identified by an ability of demineralized bone extract to induce endochondral osteogenesis in vivo in an extraskeletal site.
Function
[編集]BMP4は、TGF-βスーパーファミリーに属するポリペプチドである。他の骨形態形成タンパク質と同様に、骨と軟骨の発生、特に歯と四肢の発生と骨折修復に関与している。この特定のファミリーメンバーは、ヒトにおける軟骨内骨形成の開始において重要な役割を果たしている。筋肉の発達、骨ミネラル化、尿管芽の発達にも関与していることが示されている[4]。
BMP4 is a polypeptide belonging to the TGF-β superfamily of proteins. It, like other bone morphogenetic proteins, is involved in bone and cartilage development, specifically tooth and limb development and fracture repair. This particular family member plays an important role in the onset of endochondral bone formation in humans. It has been shown to be involved in muscle development, bone mineralization, and ureteric bud development.[4]
BMP4は、その上の外胚葉組織の分化を刺激する。[5]
BMP4 stimulates differentiation of overlying ectodermal tissue. [5]
骨形成タンパク質は、成体動物の骨形成を刺激することが知られている。要出典】BMPは胚発生において大きな役割を果たすことが知られている。胚では、BMP4は腹側中胚葉を誘導することにより、Xenopusカエルの背腹軸形成に役立っている。マウスでは、BMP4の標的化不活性化によって中胚葉の形成が阻害される[6]。また、BMP7の助けを借りて発達中の神経管の背側-腹側パターニングを確立し、背側文字を誘導する。
Bone morphogenetic proteins are known to stimulate bone formation in adult animals. This is thought that inducing osteoblastic commitment and differentiation of stem cells such as mesenchymal stem cells.[要出典]BMPs are known to play a large role in embryonic development. In the embryo BMP4 helps establish dorsal-ventral axis formation in Xenopus frog through inducing ventral mesoderm. In mice targeted inactivation of BMP4 disrupts mesoderm from forming.[6] As well establishes dorsal-ventral patterning of the developing neural tube with the help of BMP7, and inducing dorsal characters.
BMP4はまた、神経組織ではなく表皮の形成を誘導することによって、ゼノプス胚における神経分化の程度を制限する。BMP4は体節における側性の誘導を助けることができる。体節は軟骨、骨、胴体背側の真皮、胸部の筋肉、四肢の筋肉の発生に必要である[7]。BMP4は神経堤細胞のアポトーシスを誘導することで、発達中の頭部のパターニングを助けるが、これは後脳で行われる。[8]
BMP4 also limits the extent to which neural differentiation in Xenopus embryos occurs by inducing epidermis formation rather than neural tissue. They can aid in inducing the lateral characteristics in somites. Somites are required for the development of cartilage, bone, dermis on the dorsal side of the body, thoracic muscles and muscles within limbs.[7] BMP4 helps in the patterning of the developing head though inducing apoptosis of the neural crest cells; this is done in the hindbrain.
成体では、BMP4は脳の2つの神経発生ニッチ、海馬歯状回と側脳室に隣接する脳室下帯(SVZ)で生涯を通じて起こる神経発生(すなわち、新しいニューロンの生成)に重要である。これらのニッチでは、新しいニューロンが幹細胞から絶えず生成されている。実際、歯状回ではBMP4が神経幹細胞を静止状態に維持し、幹細胞プールの枯渇を防いでいることが示されている[9]。SVZでは、成体神経幹細胞からの神経新生を開始し、乏突起膠新生の代替運命を抑制するために、Smad4を介したBMP媒介シグナル伝達が必要である。[10]さらに、SVZではBMP4が分化促進作用を持つことが示されている。BMP4は、終末分化に必要な遺伝子Tis21/BTG2が欠損したSVZ神経球における終末分化の欠損を救うからである[11]。Tis21はSVZにおけるBMP4発現の正の制御因子である[11]。
In adult, BMP4 is important for the neurogenesis (i.e., the generation of new neurons) that occurs throughout life in two neurogenic niches of the brain, the dentate gyrus of the hippocampus and the subventricular zone (SVZ) adjacent to lateral ventricles. In these niches new neurons are continuously generated from stem cells. In fact it has been shown that in the dentate gyrus BMP4 maintains neural stem cells in quiescence, thus preventing the depletion of the pool of stem cells.[8] In the SVZ , BMP-mediated signaling via Smad4 is required to initiate neurogenesis from adult neural stem cells and suppress the alternative fate of oligodendrogliogenesis.[9] Moreover, it has been shown that in the SVZ BMP4 has a prodifferentiative effect, since it rescues a defect of terminal differentiation in SVZ neurospheres where the gene Tis21/BTG2 - required for terminal differentiation - has been deleted.[10] Tis21 is a positive regulator of BMP4 expression in the SVZ.[10]
BMP4は骨と軟骨の代謝に重要である。BMP4シグナル伝達は、初期中胚葉と生殖細胞の形成に見られる。四肢芽の制御、肺、肝臓、歯、顔面間充織細胞の発生は、BMP4シグナルに起因する他の重要な機能である。[12] 趾形成は、他のBMPシグナルとともにBMP4の影響を受ける。趾間充織はBMP4を発現し、この領域のアポトーシスを防ぐ[13]。これらの転写因子は形成された歯を切歯にする。
BMP4 is important for bone and cartilage metabolism. The BMP4 signaling has been found in formation of early mesoderm and germ cells. Limb bud regulation and development of the lungs, liver, teeth and facial mesenchyme cells are other important functions attributed to BMP4 signaling.[11] Digit formation is influenced by BMP4, along with other BMP signals. The interdigital mesenchyme exhibits BMP4, which prevents apoptosis of the region.[12] Tooth formation relies on BMP4 expression, which induces Msx 1 and 2. These transcription factors turn the forming tooth to become and incisor.
BMP4はまた、脂肪組織においても重要な役割を果たしている。白色脂肪の形成に必須であり、脂肪細胞の分化を促進する[14]。さらに、褐色脂肪にとっても重要であり、褐色脂肪ではUCP1を誘導し、非震盪性熱発生に関連する[14]。
BMP4 also plays important roles in adipose tissue: it is essential for white adipogenesis, and promotes adipocyte differentiation.[13] Additionally, it is also important for brown fat, where it induces UCP1, related to non-shivering thermogenesis.[13]
BMP4 secretion helps cause differentiation of the ureteric bud into the ureter.[14]
BMP4 antagonizes organizer tissue and is expressed in early development in ectoderm and mesoderm tissue. Upon gastrulation, the transcription of BMP4 is limited to the ventrolateral marginal zone due to inhibition from the dorsalizing side of the developing embryo. BMP4 aids in ventralizing mesoderm, which guides the dorsal-ventral axis formation. In Xenopus BMP4 has been found to aid in formation of blood and blood islands. [15]
BMP4, initially expressed in the epidermis, is found in the roof plate during formation of the neural tube. A gradient of BMP signaling is found in opposition to a Sonic hedgehog, Shh, gradient. This expression of BMP4 patterns the dorsal neurons.[16]
BMP4, in conjunction with FGF2, promote differentiation of stem cells to mesodermal lineages. After differentiation, BMP4 and FGF2 treated cells generally produces higher amounts of osteogenic and chondrogenic differentiation than untreated stem cells.[17] Also in conjunction with FGF2 it can produce progenitor thyroid cells from pluripotent stem cells in mice and humans.[18]
BMP4 has been shown to induce the expression of the Msx gene family, which is believed to be part of cartilage formation from somitic mesoderm.[19]
BMP4, a paracrine growth factor, has been found in rat ovaries. BMP4, in conjunction with BMP7, regulate early ovarian follicle development and primordial-to-primary follicle transition. In addition, inhibition of BMP4 with antibodies has been shown to decrease overall ovary size. These results indicate that BMP4 may aid in survival and prevention of apoptosis in oocytes.[11]
In birds, BMP4 has been shown to influence the beak size of Darwin's finches. Low amounts of BMP4 are correlated with low beak depths and widths. Conversely, high BMP4 expression makes high beak depths and widths. The genetic regulation of BMP4 provides the foundation for natural selection in bird beaks.[20]
Protein structure
[編集]Yielding an active carboxy-terminal peptide of 116 residues, human bmp4 is initially synthesized as a forty percent residue preproprotein which is cleaved post translationally. BMP4 has seven residues which are conserved and glycosylated.[21] The monomers are held with disulphide bridges and 3 pairs of cysteine amino acids. This conformation is called a “cystine knot”. BMP4 can form homodimers or heterodimers with similar BMPS. One example of this is BMP7. This ability to form homodimers or heterodimers gives the ability to have greater osteoinductive activity than just bmp4 alone.[22] Not much is known yet about how BMPS interact with the extracellular matrix. As well little is known about the pathways which then degrade BMP4.
Inhibition
[編集]Inhibition of the BMP4 signal (by chordin, noggin, or follistatin) causes the ectoderm to differentiate into the neural plate. If these cells also receive signals from FGF, they will differentiate into the spinal cord; in the absence of FGF the cells become brain tissue.
While overexpression of BMP4 expression can lead to ventralization, inhibition with a dominant negative may result in complete dorsalization of the embryo or the formation of two axises.[23]
It is important to note that mice in which BMP4 was completely inactivated usually died during gastrulation. It is thought that inactivation of human BMP4 would likely have the same effect. However, mutations which don't entirely inactivate BMP4 in humans can also have subtle effects phenotypically, and have been implicated in tooth agenesis as well as osteoporosis.[24]
Isoforms
[編集]Alternative splicing in the 5' untranslated region of this gene has been described and three variants are described, all encoding an identical protein.[25]
Molecular mechanisms
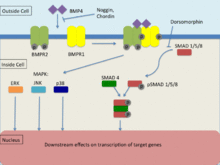
[編集]BMP4, as a member of the transforming growth factor-β (TGF-β) family binds to 2 different types of serine-threonine kinase receptors known as BMPR1 and BMPR2.[26] Signal transduction via these receptors occurs via Smad and map kinase pathways to effect transcription of its target genes. In order for signal transduction to occur, both receptors must be functional. BMP is able to bind to BMPR2 without BMPR1 however, the affinity significantly increases in the presence of both receptors. BMPR1 is transphosphorylated via BMPR2 which induces downstream signalling within the cell, affecting transcription.[26]
Smad signaling pathway
[編集]TGF-β family receptors most commonly use the Smad signaling pathway to tranduce signals.[26] Type 2 receptors are responsible for activating type 1 receptors where their function involves the phosphorylation of R-Smads (Smad-1, Smad-5, Smad-8). Upon phosphorylation, formation of an R-SMAD complex in conjunction with common-partner Smad (co-Smad) occurs where it migrates to the nucleus. This signaling pathway is regulated by the small molecule inhibitor known as dorsomorphin which prevents the downstream effects of R-smads.[26]
Map kinase (MAPK) signaling pathways
[編集]Mitogen activated protein kinases (MAPK) undergo phosphorylation via a signaling cascade where MAPKKK phosphorylates and activates MAPKK and MAPKK phosphorylates and activates MAPK which then induces an intracellular response.[27] Activation of MAPKKK is through the interaction of mainly GTPases or another group of protein kinases. TGF-β receptors induce the MAPK signaling pathways of ERK, JNK and p38.[27] BMP4 is also known to activate the ERK, JNK and p38 MAPK signalling pathways whilst have been found to act independently of Smad signaling pathways, are mostly active in conjunction with Smad.[28] The activation of the ERK and JNK pathways acts to phosphorylate Smad and therefore regulate its activation. In addition to this, MAPK pathways may be able to directly affect Smad-interacting transcription factors via a JNK or p38 substrate that induces convergence of the two signaling pathways. This convergence is noted to consist mainly of cooperative behavior however, there is evidence to suggest that they may at times counteract each other. Furthermore, the balance that exists between the direct activation of these signaling pathways has a significant effect on TGF-β induced cellular responses.[28]
Clinical significance
[編集]Increase in expression of BMP4 has been associated with a variety of bone diseases, including the heritable disorder Fibrodysplasia Ossificans Progressiva.[29]
There is strong evidence from sequencing studies of candidate genes involved in clefting that mutations in the bone morphogenetic protein 4 (BMP4) gene may be associated in the pathogenesis of cleft lip and palate.[30]
Eye development
[編集]Eyes are essential for organisms, especially terrestrial vertebrates, to observe prey and obstacles; this is critical for their survival. The formation of the eyes starts as optic vesicles and lens derived from the neuroectoderm. Bone morphogenic proteins are known to stimulate eye lens formation. During early development of eyes, the formation of the optic vesicle is essential in Mice and BMP4 expressed strongly in the optic vesicle and weakly in the surrounding mesenchyme and surface ectoderm. This concentration gradient of BMP4 in optic vesicle is critical for lens induction. Researcher, Dr. Furuta and Dr. Hogan found out that if they did a laser mutation on mice embryos and causing a BMP4 homozygous null mutation, this embryo will not develop the lens. They also did an in situ hybridization of the BMP4 gene showing green color and Sox2 gene in red which they thought it was involved in the lens formation as well. After they did these two in situ hybridizations in the mice embryos, they found that both green and red colors are found in the optic vesicle of the mice embryos. This indicated that BMP4 and Sox2 are expressed in the right place at the right time of the optic vesicle and prove that they have some essential functions for the lens induction. Furthermore, they did a follow-up experiment that by injecting BMP4 into the BMP4 homozygous mutant embryos rescued the lens formation (12). This indicated that BMP4 is definitely required for lens formation. However, researchers also found that some of the mutated mice cannot be rescued. They later found that those mutants lacked of Msx 2 which is activated by BMP4. The mechanism they predicted was that BMP4 will active Msx 2 in the optic vesicle and concentration combination of BMP4 and Msx2 together active Sox2 and the Sox2 is essential for lens differentiation.[31]
Injection of Noggin into lens fiber cells in mice significantly reduces the BMP4 proteins in the cells. This indicates that Noggin is sufficient to inhibit the production of BMP4. Moreover, another inhibitor protein, Alk6 was found that blocked the BMP4 from activating the Msx2 which stopped lens differentiation .[32] However, there are still a lot of unknown about the mechanism of inhibition on BMP4 and downstream regulation of Sox2. In the future, researchers are aiming to find out a more complete pathway of whole eye development and hoping one day, they can find a way to cure some genetic caused eye diseases.
Hair loss
[編集]Hair loss or known as alopecia is caused from the changing of hair follicle morphology and hair follicle cycling in an abnormal fashion.[33] The cycles of hair follicles are that of growth, or anagen, regression or catagen, and rest or telogen.[34] In mammals reciprocal epithelial and mesenchymal interactions control the development of hair. Genes such as BMP4 and BMP2 are both active within the precursors of the hair shaft. Specifically BMP4 is found in the dermal papilla. BMP4 is part of the signaling network which controls the development of hair. It is needed for the induction of biochemical pathways and signaling for regulating the differentiation of the hair shaft in the anagen hair follicle. This is done through controlling the expression of the transcription factors which regulate hair differentiation. It is still unclear however where BMPs act within the genetic network. The signaling of bmp4 may potentially control expression of terminal differentiation molecules such as keratins. Other regulators have been shown to control hair follicle development as well. HOXC13 and FOXN1 are considered important regulators because loss-of-function experiments show impaired hair shaft differentiation that doesn't interfere in the hair follicle formation.[35]
When BMP4 is expressed ectopically, within transgenic mice the hair follicle outer root sheath (ORS) the proliferation of the cell matrix is inhibited. BMP4 also activates hair keratin gene expression noting that BMP4 is important in the differentiation of the hair shaft. Noggin, a known inhibitor of BMP4, is found within the matrix cells of the hair bulb. Other important factors to consider in the development of hair is the expression of Shh (sonic hedgehog), BMP7, BMP2, WNT, and β-catenin as these are required in early stage morphogenesis.[36]
Other genes which can inhibit or interact with BMP4 are noggin, follistatin, gremlin, which is all expressed in the developing hair follicles.[37] In mice in which noggin is lacking, there are fewer hair follicles than on a normal mouse and the development of the follicle is inhibited. In chick embryos it is shown that ectopically expressed noggin produces enlarged follicles, and BMP4 signaling shows repressed placode fate in nearby cells.[22] Noggin has also been shown during in vivo experiments to induce hair growth in post natal skin.[38]
BMP4 is an important component of the biological pathways that involved regulating hair shaft differentiation within the anagen hair follicle. The strongest levels of expressed BMP4 are found within the medulla, hair shaft cells, distal hair matrix, and potential precursors of the cuticle. The two main methods which BMP4 inhibit expression of hair is through restricting growth factor expression in the hair matrix and antagonism between growth and differentiation signaling.[36]
Pathways that regulate hair follicle formation and hair growth are key in developing therapeutic methods for hair loss conditions. Such conditions include the development of new follicles, changing the shape of characteristics of existing follicles, and the altering of hair growth in existing hair follicles. Furthermore, BMP4 and the pathway through which it works may provide therapeutic targets for the prevention of hair loss.[34]
References
[編集]Further reading
[編集]
- “Novel regulators of bone formation: molecular clones and activities”. Science 242 (4885): 1528–34. (1989). Bibcode: 1988Sci...242.1528W. doi:10.1126/science.3201241. PMID 3201241.
- “Cloning and characterization of a human type II receptor for bone morphogenetic proteins”. Proc. Natl. Acad. Sci. U.S.A. 92 (17): 7632–6. (1995). Bibcode: 1995PNAS...92.7632R. doi:10.1073/pnas.92.17.7632. PMC 41199. PMID 7644468.
- “Identification of a human type II receptor for bone morphogenetic protein-4 that forms differential heteromeric complexes with bone morphogenetic protein type I receptors”. J. Biol. Chem. 270 (38): 22522–6. (1995). doi:10.1074/jbc.270.38.22522. PMID 7673243.
- “A mammalian serine/threonine kinase receptor specifically binds BMP-2 and BMP-4”. Biochem. Biophys. Res. Commun. 205 (3): 1944–51. (1995). doi:10.1006/bbrc.1994.2898. PMID 7811286.
- “Expression of bone morphogenetic protein messenger RNAs by normal rat and human prostate and prostate cancer cells”. Prostate 24 (4): 204–11. (1994). doi:10.1002/pros.2990240406. PMID 8146069.
- “Genomic organization of the human bone morphogenetic protein-4 gene: molecular basis for multiple transcripts”. Biochem. Biophys. Res. Commun. 219 (3): 789–94. (1996). doi:10.1006/bbrc.1996.0312. hdl:2066/23948. PMID 8645259.
- “Identification of type I and type II serine/threonine kinase receptors for growth/differentiation factor-5”. J. Biol. Chem. 271 (35): 21345–52. (1996). doi:10.1074/jbc.271.35.21345. PMID 8702914.
- “Normalization and subtraction: two approaches to facilitate gene discovery”. Genome Res. 6 (9): 791–806. (1997). doi:10.1101/gr.6.9.791. PMID 8889548.
- “The human bone morphogenetic protein 4 (BMP-4) gene: molecular structure and transcriptional regulation”. Calcif. Tissue Int. 63 (3): 221–9. (1998). doi:10.1007/s002239900518. PMID 9701626.
- “Transformation of tooth type induced by inhibition of BMP signaling”. Science 282 (5391): 1136–8. (1998). Bibcode: 1998Sci...282.1136T. doi:10.1126/science.282.5391.1136. PMID 9804553.
- “Functional characterization of two promoters in the human bone morphogenetic protein-4 gene”. J. Bone Miner. Res. 14 (8): 1432–41. (1999). doi:10.1359/jbmr.1999.14.8.1432. PMID 10457277.
- “Noggin is a negative regulator of neuronal differentiation in developing neocortex”. Dev. Neurosci. 22 (1–2): 68–73. (2000). doi:10.1159/000017428. PMID 10657699.
- “BMP-4 affects the differentiation of metanephric mesenchyme and reveals an early anterior-posterior axis of the embryonic kidney”. Dev. Dyn. 217 (2): 146–58. (2000). doi:10.1002/(SICI)1097-0177(200002)217:2<146::AID-DVDY2>3.0.CO;2-I. PMID 10706139.
- “Antiestrogens specifically up-regulate bone morphogenetic protein-4 promoter activity in human osteoblastic cells”. Mol. Endocrinol. 14 (5): 623–33. (2000). doi:10.1210/mend.14.5.0463. PMID 10809227.
- “Requirement of Bmp8b for the generation of primordial germ cells in the mouse”. Mol. Endocrinol. 14 (7): 1053–63. (2000). doi:10.1210/mend.14.7.0479. PMID 10894154.
- “Effect of extracellular calcium on the gene expression of bone morphogenetic protein-2 and -4 of normal human bone cells”. J. Bone Miner. Metab. 19 (1): 13–9. (2001). doi:10.1007/s007740170055. PMID 11156467.
- “Identification of the ligand-binding site of the BMP type IA receptor for BMP-4”. Biopolymers 55 (5): 399–406. (2001). doi:10.1002/1097-0282(2000)55:5<399::AID-BIP1014>3.0.CO;2-9. PMID 11241215.
- “Synergistic effects of different bone morphogenetic protein type I receptors on alkaline phosphatase induction”. J. Cell Sci. 114 (Pt 8): 1483–9. (2001). doi:10.1242/jcs.114.8.1483. PMID 11282024.
- “Development of Axon-Target Specificity of Ponto-Cerebellar Afferents”. PLOS Biology 9 (2): e1001013. (2011). doi:10.1371/journal.pbio.1001013. PMC 3035609. PMID 21346800.
- “Towards a molecular understanding of hair loss and its treatment”. Trends Mol Med 7 (7): 293–301. (July 2001). doi:10.1016/S1471-4914(01)02027-5. PMID 11425637.
- “Expression of activin subunits, activin receptors and follistatin in postimplantation mouse embryos suggests specific developmental functions for different activins”. Development 120 (12): 3621–37. (December 1994). doi:10.1242/dev.120.12.3621. PMID 7821227.
- “The signalling molecule BMP4 mediates apoptosis in the rhombencephalic neural crest”. Nature 372 (6507): 684–6. (December 1994). Bibcode: 1994Natur.372..684G. doi:10.1038/372684a0. PMID 7990961.
- “beta-Catenin controls hair follicle morphogenesis and stem cell differentiation in the skin”. Cell 105 (4): 533–45. (May 2001). doi:10.1016/S0092-8674(01)00336-1. PMID 11371349.
- “Inhibition of Bmp signaling affects growth and differentiation in the anagen hair follicle”. EMBO J. 19 (24): 6664–74. (December 2000). doi:10.1093/emboj/19.24.6664. PMC 305899. PMID 11118201.
- “Bone morphogenic protein-4”. Int. J. Biochem. Cell Biol. 28 (12): 1293–6. (December 1996). doi:10.1016/S1357-2725(96)00075-1. PMID 9022288.
- “Dorsal differentiation of neural plate cells induced by BMP-mediated signals from epidermal ectoderm”. Cell 82 (6): 969–79. (September 1995). doi:10.1016/0092-8674(95)90276-7. PMID 7553857.
- “Molecular mechanisms regulating hair follicle development”. J. Invest. Dermatol. 118 (2): 216–25. (February 2002). doi:10.1046/j.0022-202x.2001.01670.x. PMID 11841536.
- “Lateral and axial signals involved in avian somite patterning: a role for BMP4”. Cell 84 (3): 461–71. (February 1996). doi:10.1016/S0092-8674(00)81291-X. PMID 8608600.
- “Bone morphogenetic protein-2 causes commitment and differentiation in C3H10T1/2 and 3T3 cells”. Growth Factors 9 (1): 57–71. (1993). doi:10.3109/08977199308991582. PMID 8347351.
- “Bone morphogenetic protein-4 is required for mesoderm formation and patterning in the mouse”. Genes Dev. 9 (17): 2105–16. (September 1995). doi:10.1101/gad.9.17.2105. PMID 7657163.
External links
[編集]- BMPedia - the Bone Morphogenetic Protein Wiki[リンク切れ]
- BMP4 human gene location in the UCSC Genome Browser.
- BMP4 human gene details in the UCSC Genome Browser.
Template:TGF beta signalingTemplate:TGFβ receptor superfamily modulators
[[Category:映像がある記事]] [[Category:未査読の翻訳があるページ]]
- ^ “Fine mapping of the human bone morphogenetic protein-4 gene (BMP4) to chromosome 14q22-q23 by in situ hybridization”. Genomics 27 (3): 559–60. (November 1995). doi:10.1006/geno.1995.1096. hdl:2066/22049. PMID 7558046.
- ^ “Cloning and sequence of bone morphogenetic protein 4 (BMP-4) from a human placental cDNA library”. DNA Seq 5 (5): 273–5. (November 1995). doi:10.3109/10425179509030980. PMID 7579580.
- ^ “Structure and expression of Xenopus tropicalis BMP-2 and BMP-4 genes”. Mech. Dev. 109 (1): 79–82. (November 2001). doi:10.1016/S0925-4773(01)00506-8. PMID 11677055.
- ^ “Bone morphogenetic protein 4 regulates the budding site and elongation of the mouse ureter”. The Journal of Clinical Investigation 105 (7): 863–873. (April 2000). doi:10.1172/JCI8256. PMID 10749566.
- ^ “The differentiation effect of bone morphogenetic protein (BMP) on human amniotic epithelial stem cells to express ectodermal lineage markers”. Cell and Tissue Research 383 (2): 751–763. (February 2021). doi:10.1007/s00441-020-03280-z. PMID 32960356.
- ^ “Bone morphogenetic protein-4 is required for mesoderm formation and patterning in the mouse”. Genes & Development 9 (17): 2105–2116. (September 1995). doi:10.1101/gad.9.17.2105. PMID 7657163.
- ^ Wolpert, Lewis; Tickle, Cheryl; Arias, Alfonso Martinez; Lawrence, Peter; Lumsden, Andrew; Robertson, Elizabeth; Meyerowitz, Elliot; Smith, Jim (2015). “Vertebrate development III: Chick and mouse - completing the body plan”. Principles of development (Fifth ed.). Oxford, United Kingdom. p. 207. ISBN 9780198709886
- ^ “Signaling through BMPR-IA regulates quiescence and long-term activity of neural stem cells in the adult hippocampus”. Cell Stem Cell 7 (1): 78–89. (2010). doi:10.1016/j.stem.2010.04.016. PMID 20621052.
- ^ “Adult neurogenesis requires Smad4-mediated bone morphogenic protein signaling in stem cells”. The Journal of Neuroscience 28 (2): 434–446. (2008). doi:10.1523/JNEUROSCI.4374-07.2008. PMC 6670509. PMID 18184786.
- ^ a b “Tis21 is required for adult neurogenesis in the subventricular zone and for olfactory behavior regulating cyclins, BMP4, Hes1/5 and Ids”. Front Cell Neurosci 8: 98. (2014). doi:10.3389/fncel.2014.00098. PMC 3977348. PMID 24744701.
- ^ a b “Bone morphogenetic protein-4 acts as an ovarian follicle survival factor and promotes primordial follicle development”. Biol. Reprod. 69 (4): 1265–72. (October 2003). doi:10.1095/biolreprod.103.018671. PMID 12801979.
- ^ “Regulation of limb patterning by extracellular microfibrils”. J. Cell Biol. 154 (2): 275–81. (July 2001). doi:10.1083/jcb.200105046. PMC 2150751. PMID 11470817.
- ^ a b “Beyond the bone: Bone morphogenetic protein signaling in adipose tissue”. Obesity Reviews 20 (5): 648–658. (May 2019). doi:10.1111/obr.12822. PMC 6447448. PMID 30609449.
- ^ “Evidence that bone morphogenetic protein 4 has multiple biological functions during kidney and urinary tract development”. Kidney Int. 63 (3): 835–44. (March 2003). doi:10.1046/j.1523-1755.2003.00834.x. PMID 12631064.
- ^ “Ventral mesodermal patterning in Xenopus embryos: expression patterns and activities of BMP-2 and BMP-4”. Dev. Genet. 17 (1): 78–89. (1995). doi:10.1002/dvg.1020170109. PMID 7554498.
- ^ “Effects of Shh and Noggin on neural crest formation demonstrate that BMP is required in the neural tube but not ectoderm”. Development 125 (24): 4919–30. (December 1998). doi:10.1242/dev.125.24.4919. PMID 9811576.
- ^ “Enhancement of osteogenic and chondrogenic differentiation of human embryonic stem cells by mesodermal lineage induction with BMP-4 and FGF2 treatment”. Biochem. Biophys. Res. Commun. 430 (2): 793–7. (January 2013). doi:10.1016/j.bbrc.2012.11.067. PMID 23206696.
- ^ “Regeneration of Thyroid Function by Transplantation of Differentiated Pluripotent Stem Cells” (英語). Cell Stem Cell 17 (5): 527–42. (November 2015). doi:10.1016/j.stem.2015.09.004. PMC 4666682. PMID 26593959.
- ^ “A role for BMP-4 in the development of subcutaneous cartilage”. Mech. Dev. 57 (1): 69–78. (June 1996). doi:10.1016/0925-4773(96)00534-5. PMID 8817454.
- ^ “Bmp4 and morphological variation of beaks in Darwin's finches”. Science 305 (5689): 1462–5. (September 2004). Bibcode: 2004Sci...305.1462A. doi:10.1126/science.1098095. PMID 15353802.
- ^ “Potent ectopic bone-inducing activity of bone morphogenetic protein-4/7 heterodimer”. Biochem. Biophys. Res. Commun. 210 (3): 670–7. (May 1995). doi:10.1006/bbrc.1995.1712. PMID 7763240.
- ^ a b “Noggin is a mesenchymally derived stimulator of hair-follicle induction”. Nat. Cell Biol. 1 (3): 158–64. (July 1999). doi:10.1038/11078. PMID 10559902. 引用エラー: 無効な
<ref>タグ; name "pmid10559902"が異なる内容で複数回定義されています - ^ “Structural and functional analysis of the BMP-4 promoter in early embryos of Xenopus laevis”. Mech. Dev. 74 (1–2): 29–39. (June 1998). doi:10.1016/S0925-4773(98)00059-8. PMID 9651472.
- ^ Yu, Miao; Wang, Hao; Fan, Zhuangzhuang; Xie, Chencheng; Liu, Haochen; Liu, Yang; Han, Dong; Wong, Sing-Wai et al. (July 2019). “BMP4 mutations in tooth agenesis and low bone mass”. Archives of Oral Biology 103: 40–46. doi:10.1016/j.archoralbio.2019.05.012.
- ^ “Entrez Gene: BMP4 bone morphogenetic protein 4”. Template:Cite webの呼び出しエラー:引数 accessdate は必須です。
- ^ a b c d “Bone morphogenetic protein receptors and signal transduction”. J. Biochem. 147 (1): 35–51. (January 2010). doi:10.1093/jb/mvp148. PMID 19762341.
- ^ a b Cell Signaling Technology, Inc.. “Mitogen-Activated Protein Kinase Cascades”. 17 November 2012閲覧。
- ^ a b “Smad-dependent and Smad-independent pathways in TGF-beta family signaling”. Nature 425 (6958): 577–84. (October 2003). Bibcode: 2003Natur.425..577D. doi:10.1038/nature02006. PMID 14534577.
- ^ “Transgenic Mice Overexpressing BMP4 Develop a Fibrodysplasia Ossificans Progressiva (FOP)-Like Phenotype”. Am. J. Pathol. 165 (4): 1107–15. (October 2004). doi:10.1016/S0002-9440(10)63372-X. PMC 1618644. PMID 15466378.
- ^ “Cleft lip and palate: understanding genetic and environmental influences”. Nat. Rev. Genet. 12 (3): 167–78. (March 2011). doi:10.1038/nrg2933. PMC 3086810. PMID 21331089.
- ^ “BMP4 is essential for lens induction in the mouse embryo”. Genes Dev. 12 (23): 3764–75. (December 1998). doi:10.1101/gad.12.23.3764. PMC 317259. PMID 9851982.
- ^ “Bmp signaling is required for development of primary lens fiber cells”. Development 129 (15): 3727–37. (August 2002). doi:10.1242/dev.129.15.3727. PMID 12117821.
- ^ “Towards a molecular understanding of hair loss and its treatment”. Trends Mol Med 7 (7): 293–301. (July 2001). doi:10.1016/S1471-4914(01)02027-5. PMID 11425637.
- ^ a b “Molecular mechanisms regulating hair follicle development”. J. Invest. Dermatol. 118 (2): 216–25. (February 2002). doi:10.1046/j.0022-202x.2001.01670.x. PMID 11841536. 引用エラー: 無効な
<ref>タグ; name "Millar_2002"が異なる内容で複数回定義されています - ^ “Inhibition of Bmp signaling affects growth and differentiation in the anagen hair follicle”. EMBO J. 19 (24): 6664–74. (December 2000). doi:10.1093/emboj/19.24.6664. PMC 305899. PMID 11118201.
- ^ a b “beta-Catenin controls hair follicle morphogenesis and stem cell differentiation in the skin”. Cell 105 (4): 533–45. (May 2001). doi:10.1016/S0092-8674(01)00336-1. PMID 11371349. 引用エラー: 無効な
<ref>タグ; name "Huelsken_2001"が異なる内容で複数回定義されています - ^ “Expression of activin subunits, activin receptors and follistatin in postimplantation mouse embryos suggests specific developmental functions for different activins”. Development 120 (12): 3621–37. (December 1994). doi:10.1242/dev.120.12.3621. PMID 7821227.
- ^ “Noggin is required for induction of the hair follicle growth phase in postnatal skin”. FASEB J. 15 (12): 2205–14. (October 2001). doi:10.1096/fj.01-0207com. PMID 11641247.
